
Tests of Big Bang: The Light Elements
Nucleosynthesis in the Early Universe
The term nucleosynthesis refers to the formation of heavier elements, atomic nuclei with many protons and neutrons, from the fusion of lighter elements. The Big Bang theory predicts that the early universe was a very hot place. One second after the Big Bang, the temperature of the universe was roughly 10 billion degrees and was filled with a sea of neutrons, protons, electrons, anti-electrons (positrons), photons and neutrinos. As the universe cooled, the neutrons either decayed into protons and electrons or combined with protons to make deuterium (an isotope of hydrogen). During the first three minutes of the universe, most of the deuterium combined to make helium. Trace amounts of lithium were also produced at this time. This process of light element formation in the early universe is called “Big Bang nucleosynthesis” (BBN).
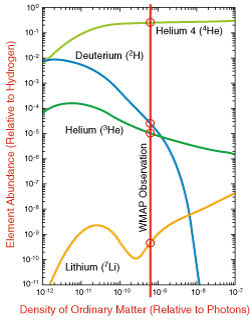 The predicted abundance of deuterium, helium and lithium depends on the
density of ordinary matter in the early universe, as
shown in the figure at left. These results indicate that the yield of helium is relatively
insensitive to the abundance of ordinary matter, above a certain threshold. We generically
expect about 24% of the ordinary matter in the universe to be helium produced in the Big
Bang. This is in very good agreement with observations and is another major triumph for the Big Bang theory.
The predicted abundance of deuterium, helium and lithium depends on the
density of ordinary matter in the early universe, as
shown in the figure at left. These results indicate that the yield of helium is relatively
insensitive to the abundance of ordinary matter, above a certain threshold. We generically
expect about 24% of the ordinary matter in the universe to be helium produced in the Big
Bang. This is in very good agreement with observations and is another major triumph for the Big Bang theory.
However, the Big Bang model can be tested further. Given a precise measurement of the abundance of ordinary matter, the predicted abundances of the other light elements becomes highly constrained. The WMAP satellite is able to directly measure the ordinary matter density and finds a value of 4.6% (±0.2%), indicated by the vertical red line in the graph. This leads to predicted abundances shown by the circles in the graph, which are in good agreement with observed abundances. This is an important and detailed test of nucleosynthesis and is further evidence in support of the Big Bang theory. Had the results been in conflict, it would point to 1) errors in the data, 2) an incomplete understanding of the process of Big Bang nucleosynthesis, 3) a misunderstanding of the mechanisms that produce fluctuations in the microwave background radiation, or 4) a more fundamental problem with the Big Bang theory.
Nucleosynthesis in Stars
Elements heavier than lithium are all synthesized in stars. During the late stages of stellar evolution, massive stars burn helium to carbon, oxygen, silicon, sulfur, and iron. Elements heavier than iron are produced in two ways: in the outer envelopes of super-giant stars and in the explosion of a supernovae. All carbon-based life on Earth is literally composed of stardust.
Some History
In 1948, Physicist George Gamow hypothesized that all of the elements might have been made in the hot and dense early universe. He suggested to his student, Ralph Alpher, that he calculate this. Alpher did so for his PhD thesis, with Robert Herman participating in much of the work. Alpher and Herman found that Gamow was wrong: most elements could not have been made in the early universe. The problem is with neutron capture. Neutrons decay in about 10 minutes, and their density decreases as the universe expands in that time. There just isn't enough time to keep building up to the heavier elements before the neutrons are gone. The heavy elements are made later, in stars. Only the lightest elements are built up in the early universe. The important prediction that the cosmic microwave background (CMB) exists, and has a blackbody spectrum with a temperature of about 5 degrees above absolute zero, was a by-product of this work.
